Victoria is 47.
She used to fall asleep easily.
Now she wakes at 3:17 a.m.—alert, heart slightly racing, mind turning over tomorrow’s to-do list.
Her doctor says her labs are “within range.”
Her cycles are irregular but still present.
Her cholesterol has crept upward.
She feels more reactive to stress than she used to.
She doesn’t feel broken.
She feels… recalibrating.
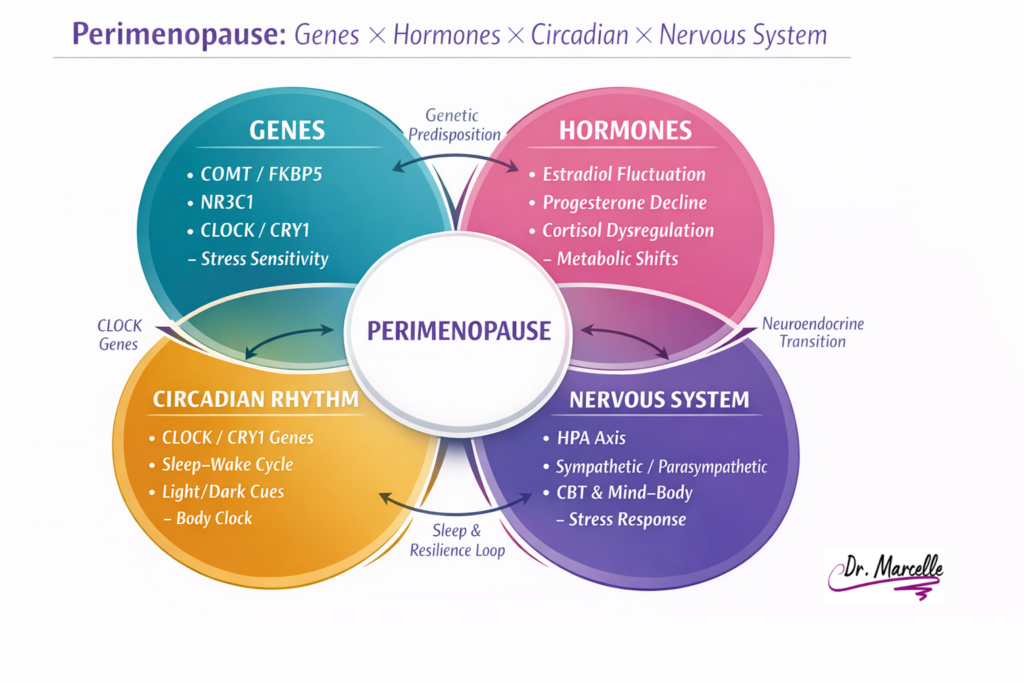
Perimenopause is not simply “low estrogen.” It is a complex neuroendocrine transition involving fluctuating estradiol, declining progesterone, altered stress signaling, sleep architecture shifts, and metabolic recalibration.¹–³
And for many women, it is the first time they truly feel how interconnected their nervous system, hormones, sleep, and resilience actually are.
This is where complementary care—when coordinated with medical care—can become the missing layer.
Not because conventional medicine is insufficient.
But because perimenopause is systemic.
The Neuroendocrine Transition: More Than Estrogen
Perimenopause is characterized not by a steady decline in estrogen, but by fluctuation. Estradiol levels can swing unpredictably before eventually declining.¹
Progesterone often declines earlier, reducing its calming, GABA-modulating influence.²
These shifts affect:
- Sleep architecture
- Mood regulation
- Vasomotor stability
- Insulin sensitivity
- Lipid metabolism
- Autonomic tone
Research shows that sleep disturbance and mood variability are significantly associated with perimenopausal transition stages.¹,³
This is not imagination. It is physiology.
But hormones do not act alone.
They operate within a regulatory network that includes the hypothalamic–pituitary–adrenal (HPA) axis, the autonomic nervous system, circadian rhythm regulators, and—in some individuals—genetic predispositions influencing stress and rhythm sensitivity.
The Stress Layer: Why Resilience Feels Different
During perimenopause, stress reactivity often changes.
Estradiol interacts with the HPA axis and influences cortisol regulation.² When estrogen fluctuates, cortisol patterns may become more reactive or dysregulated.
For some women, genetic variations may further influence this experience.
For example:
- COMT variants influence catecholamine metabolism (dopamine, epinephrine, norepinephrine). Faster or slower metabolism can affect stress perception and recovery.
- FKBP5 variants influence glucocorticoid receptor sensitivity and stress feedback regulation.
- NR3C1 relates to glucocorticoid receptor function and cortisol signaling.
These genes do not “cause” perimenopause symptoms. But they may influence how sensitive a woman is to stress load during hormonal fluctuation.
Genetics informs probability—not destiny.
And during perimenopause, probability patterns can become more visible.
The Sleep Shift: When 3:17 a.m. Becomes a Pattern
One of the most common midlife complaints is disrupted sleep.
Research shows increased sleep disturbance across the menopausal transition, even after adjusting for age.¹
Why?
Because sleep regulation depends on:
- Circadian rhythm alignment
- Melatonin signaling
- Core body temperature shifts
- Cortisol rhythm
- Autonomic stability
- Progesterone’s calming influence
When progesterone declines and estrogen fluctuates, sleep continuity can weaken.²
But there is another layer many women don’t realize:
Circadian genes.
Circadian Rhythm & the CLOCK/CRY Genes
Your circadian rhythm is governed by a transcriptional–translational feedback loop involving genes such as CLOCK, and CRY.⁴
These genes regulate 24-hour patterns in:
- Sleep–wake timing
- Cortisol rhythm
- Body temperature
- Glucose metabolism
- Inflammatory signaling
Variants in genes such as CLOCK and CRY1 have been associated with altered sleep timing and circadian preference.⁵,⁶
For example:
- Certain CLOCK polymorphisms are associated with evening chronotype tendencies and metabolic variation.⁵
- CRY1 variants have been linked with delayed sleep phase tendencies in some populations.⁶
During perimenopause, when hormonal signaling becomes less stabilizing, circadian vulnerabilities may become more apparent.
A woman who once tolerated late nights may suddenly find:
- Delayed sleep onset
- Early morning awakening
- Increased nighttime rumination
- Greater sensitivity to light exposure
- Stronger metabolic shifts with sleep loss
This is not weakness.
It is the intersection of hormonal transition and circadian regulation.
Complementary care can support:
- Light timing
- Evening wind-down physiology
- Morning cortisol anchoring
- Nutritional timing
- Nervous system regulation
- Sleep architecture stabilization
All coordinated alongside medical guidance.
The Metabolic & Cardiovascular Layer
Perimenopause is associated with shifts in lipid profile, body composition, and insulin sensitivity.³,⁷
Estradiol plays roles in:
- Nitric oxide production
- Vascular tone
- Lipid metabolism
- Fat distribution
As estrogen declines, LDL cholesterol and ApoB often rise, visceral fat may increase, and insulin sensitivity can shift.³,⁷
Sleep disruption compounds this.
Short sleep duration has been associated with adverse metabolic outcomes, including insulin resistance and cardiometabolic risk factors.⁸
This is where complementary care becomes practical—not abstract.
Supporting:
- Resistance training
- Protein adequacy
- Circadian alignment
- Stress reduction
- Sleep stabilization
can influence cardiometabolic trajectory.
Not instead of medical care—but alongside it.
Why “Normal Labs” Don’t Mean “Nothing Is Happening”
Many women in perimenopause are told their labs are normal.
That may be true.
But labs are snapshots.
Perimenopause is dynamic.
Hormone fluctuations are episodic.
Circadian rhythm shifts are patterned.
Stress responses are contextual.
Complementary care creates space to track patterns—not just numbers.
It asks:
- When do symptoms worsen?
- How is sleep structured?
- What is the stress load?
- What is the light exposure pattern?
- How consistent is meal timing?
- What is the resilience baseline?
This is systems thinking.
The Nervous System Recalibration
The autonomic nervous system (sympathetic and parasympathetic balance) plays a role in sleep, digestion, mood, and inflammatory signaling.
Mind–body interventions and Cognitive Behavioural Therapy for Insomnia (CBT-I) have demonstrated benefit in improving sleep outcomes.⁹,¹⁰
These are not “soft” interventions.
They are regulation tools.
For women with genetic tendencies toward higher stress reactivity (for example, certain FKBP5 or COMT variants), nervous system support can be particularly impactful—not because genes are destiny, but because regulation becomes more strategic.
Complementary care offers:
- Structured nervous system techniques
- Circadian stabilization strategies
- Stress physiology support
- Personalized pacing
All coordinated with medical decisions about hormone therapy, lipid management, or other interventions.
The Empowerment Layer: What You Can Do Now
If you are in perimenopause and noticing:
- Sleep fragmentation
- Mood variability
- Increased stress sensitivity
- Rising cholesterol
- Reduced recovery capacity
Consider:
- Morning light exposure within 30 minutes of waking
- Consistent sleep–wake timing
- Resistance training 2–3 times weekly
- Evening light reduction
- Protein distribution across the day
- Stress-regulation practices that feel sustainable
These are not fads. They are circadian anchors.
Complementary Care as a Missing Piece
Complementary care matters in perimenopause because:
- It validates lived experience
- It integrates nervous system regulation
- It considers circadian biology
- It incorporates genetic context without determinism
- It coordinates with medical monitoring
It does not replace diagnosis.
It does not replace hormone therapy when indicated.
It does not promise reversal.
It supports recalibration.
An Invitation
If you are navigating perimenopause and feel the shift in sleep, stress tolerance, or metabolic patterns, you are not imagining it.
Your system is recalibrating.
And sometimes, what makes the difference is not another test—but a structured, coordinated, whole-person approach that includes:
- Hormonal context
- Circadian alignment
- Nervous system regulation
- Personalized strategy
If that feels aligned, you are welcome to explore my approach at DrMarcelle.ca.
Transitions are not breakdowns.
They are recalibrations.
Warmly,
Dr. Marcelle
Health & Wellness Coach
References
- Harlow SD, et al. Executive summary of the Stages of Reproductive Aging Workshop (STRAW+10). Menopause. 2012;19(4):387–395.
- Gordon JL, Girdler SS. Hormone fluctuations and stress sensitivity during perimenopause. Psychoneuroendocrinology. 2014;50:123–136.
- Thurston RC, et al. Menopausal symptoms and cardiovascular risk. Circulation. 2019;140(7):e506–e532.
- Takahashi JS. Transcriptional architecture of the mammalian circadian clock. Nat Rev Genet. 2017;18(3):164–179.
- Garaulet M, et al. CLOCK gene polymorphisms and metabolic syndrome. Int J Obes. 2010;34(3):516–524.
- Patke A, et al. Mutation of the human circadian clock gene CRY1 in delayed sleep phase disorder. Cell. 2017;169(2):203–215.
- El Khoudary SR, et al. Menopause transition and cardiovascular disease risk. J Am Coll Cardiol. 2020;75(16):1971–1982.
- Spiegel K, et al. Sleep loss and metabolic regulation. Lancet. 1999;354(9188):1435–1439.
- Trauer JM, et al. Cognitive behavioural therapy for chronic insomnia: systematic review and meta-analysis. Ann Intern Med. 2015;163(3):191–204.
- Irwin MR, et al. Mind-body interventions and sleep outcomes. J Clin Sleep Med. 2017;13(2):307–318.





